information
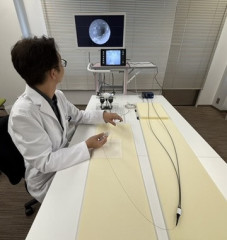
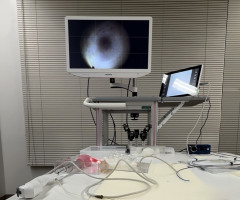
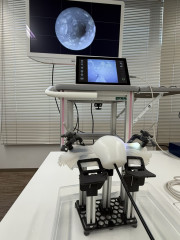
Tokyo, Japan – March 12th, 2026 – infuse Life Science Co., Ltd. announced that it has completed the prototype development of a catheter for infertility treatment under the “Medical Device Industry Entry Promotion Subsidy Program” administered by the Tokyo Metropolitan Small and Medium Enterprise Support Center.
The project focused on exploring a new design approach aimed at simplifying mechanisms that are often complex in existing therapeutic medical devices. By reducing structural complexity, the prototype is designed to improve usability for clinicians and may enable certain procedures to be performed in outpatient clinical settings.
While specific technical details are not disclosed at this stage due to ongoing intellectual property considerations, the successful completion of the prototype represents an important milestone toward further development, validation, and potential commercialization.
infuse Life Science believes that simplifying device architecture while maintaining clinical functionality may contribute to improving accessibility and efficiency in reproductive healthcare.
The company will continue advancing research and development in collaboration with clinical partners and industry stakeholders, aiming to bring innovative life science technologies to the field of reproductive medicine.
This development project was supported by the Tokyo Metropolitan Small and Medium Enterprise Support Center through its Medical Device Industry Entry Promotion Subsidy Program.
Related Grant Program
Tokyo Metropolitan Small and Medium Enterprise Support Center
Medical Device Industry Entry Promotion Subsidy Program
https://www.tokyo-kosha.or.jp/support/josei/medical/r7medical20.html
Company
Infuse Life Science Co., Ltd.
Tokyo, Japan
https://infuselifescience.com/
Forward-Looking Statements: This press release contains forward-looking statements regarding future plans and expectations. Actual results may differ due to various factors. The device has not been approved for sales by the authorities yet.
東京都中小企業振興公社「医療機器産業参入促進助成事業」における
不妊治療用カテーテル試作開発完了のお知らせ
インフューズライフサイエンス株式会社は、公益財団法人東京都中小企業振興公社が実施する「医療機器産業参入促進助成事業」の支援を受けて進めてきた不妊治療用カテーテルの試作開発を、このたび完了いたしました。
本プロジェクトでは、不妊治療に用いられる医療機器において既存技術では複雑になりがちな機構を簡略化する新しい設計アプローチの検討を行いました。これにより、医療従事者の操作性向上や、将来的には外来ベースでの治療への応用可能性が高まることが期待されます。
なお、本技術の詳細については知的財産保護の観点から現時点では公表を控えておりますが、本試作の完了は今後の開発・検証および実用化に向けた重要なマイルストーンとなります。
当社は今後も、臨床現場および産業パートナーとの連携を通じて、生殖医療分野における革新的医療技術の研究開発を進めてまいります。
将来見通しに関する注意事項
本プレスリリースには、将来の計画や期待に関する将来見通しを含む記述が含まれています。実際の結果は、さまざまな要因により異なる可能性があります。また、本製品は薬機法等未承認です。
Tokyo, Japan – January 14th, 2026 – infuse Life Science, Inc. is proud to announce the successful completion of the FY2024 Femtech Development Support and Promotion Project, a flagship initiative by the Tokyo Metropolitan Small and Medium Enterprise Support Center. Following a rigorous audit and final verification process, the company has officially received the grant funding, validating the technical maturity and operational integrity of the project.
This initiative centered on the development of a next-generation diagnostic medical device designed for the early detection of infertility causes. By providing a radiation-free, non-invasive alternative to traditional radiology-based diagnostics, infuse Life Science aims to significantly lower the barriers to early screening in reproductive health.
Dai Saito, CEO of infuse Life Science, Inc., stated:
“The successful close of this grant project is a testament to our team's efficiency and dedication to excellence. This milestone does more than just secure funding; it validates our mission to revolutionize women’s healthcare with safer, more accessible technology. As we pivot to our next growth phase—accelerating clinical trials and navigating the regulatory path toward a 2027 market launch—we are well-positioned to lead the transformation of the global femtech landscape.”
About infuse Life Science, Inc.
infuse Life Science, Inc. is a Tokyo-based startup specializing in AI-integrated medical devices for gynecology and reproductive health. Our solutions address unmet needs in infertility, oncology, and perinatal care, empowering women with safer, more accessible diagnostic tools. For more information, visit https://infuselifescience.com/ or contact us at 050-5810-2508.
Forward-Looking Statements: This press release contains forward-looking statements regarding future plans and expectations. Actual results may differ due to various factors. The device has not been approved for sales by the authorities yet.
インフューズライフサイエンス、東京都中小企業振興公社フェムテック事業を完了 革新的な不妊原因診断デバイス試作に対し助成金が正式交付
東京、日本 – 2026年1月14日 – インフューズライフサイエンス株式会社は、東京都中小企業振興公社が推進する令和6年度(2024年度)フェムテック開発支援・普及促進事業を無事完了したことを発表いたします。厳格な監査および最終検証プロセスを経て、当社は助成金の正式交付を受け、本プロジェクトの技術的成熟度と運用上の健全性を証明しました。
本事業は、不妊原因の早期発見を目的とした次世代診断医療機器の開発に焦点を当てたものです。従来の放射線を用いた診断法に代わる、放射線被ばくゼロの非侵襲型代替手段を提供することで、当社は生殖医療分野における早期スクリーニングの障壁を大幅に低減することを目指しています。
インフューズライフサイエンス株式会社 代表取締役CEO 齋藤大介のコメントです。
「この助成事業の成功裏の完了は、当社チームの効率性と卓越性への献身を物語っています。このマイルストーンは単なる資金確保にとどまらず、より安全でアクセスしやすい技術によって女性のヘルスケアに革命を起こすという私たちの使命を裏付けています。今後、臨床試験の加速と規制当局への申請を進め、2027年の市場投入を目指す次の成長フェーズに移行します。私たちは、グローバルなフェムテック分野の変革をリードする準備が整っています。」
インフューズライフサイエンス株式会社について
インフューズライフサイエンス株式会社は、東京を拠点とするスタートアップ企業で、産婦人科および生殖医療分野におけるAI統合型医療機器を専門としています。不妊、腫瘍学、周産期ケアにおける満たされていないニーズに対応し、女性に、より安全でアクセスしやすい診断ツールを提供しています。詳細は https://infuselifescience.com/ をご覧いただくか、050-5810-2508 までお問い合わせください。
将来見通しに関する注意事項
本プレスリリースには、将来の計画や期待に関する将来見通しを含む記述が含まれています。実際の結果は、さまざまな要因により異なる可能性があります。また、本製品は薬機法等未承認です。
Happy New Year 2026🎠
As we welcome the new year, we at infuse Life Science, Inc. extend our heartfelt greetings and deepest gratitude to all our stakeholders, partners, clients, and supporters for your continued trust and collaboration throughout 2025.
This past year has been one of remarkable progress and achievement across our core initiatives. In our medical device development efforts, the prototype phase for our innovative diagnostic device aimed at early detection of infertility has been successfully completed, and we look forward to announcing these exciting results through an upcoming press release. Similarly, development of our therapeutic medical device is progressing smoothly, with key achievements expected to be reported as early as January.
Our consulting business has also thrived, thanks to your invaluable support. We are proudly advancing four clients across six projects, each delivering significant outcomes and impactful results.
In the cardiovascular field, we marked a milestone by participating as a sponsor in the GREAT network meeting 2025 for the first time, which has notably increased awareness and recognition of our work among key opinion leaders in emergency and intensive care medicine in Europe and the United States.
https://www.greatnetwork.org/site/our-supporting-partners/
Reflecting on Key Advances in Reproductive Medicine in 2025
The field of reproductive medicine saw remarkable progress in 2025, with several groundbreaking developments that promise to transform diagnosis, treatment, and outcomes for patients facing infertility.
One of the most exciting advancements was the evolution of AI-driven embryo selection systems. A foundational model known as FEMI, trained on millions of time-lapse embryo images, demonstrated superior performance in predicting euploidy (chromosomal normality), blastocyst quality, and other key metrics compared to traditional manual grading and earlier AI models. This non-invasive technology achieved high accuracy in implantation potential assessment, potentially leading to improved implantation rates, higher live birth success, and reduced risks such as miscarriage—offering a more standardized and efficient approach to embryo selection in IVF cycles.
https://www.nature.com/articles/s41467-025-61116-2
Another significant breakthrough came in our understanding of polycystic ovary syndrome (PCOS), a leading cause of infertility. Research identified four distinct subtypes of PCOS—hyperandrogenic, obesity-related, high-SHBG, and high-LH/AMH—through data-driven clustering of clinical variables across large international cohorts. These subtypes differ in metabolic risks, reproductive outcomes, and complications, enabling more personalized diagnosis and treatment strategies. For instance, certain subtypes showed higher live birth rates or specific risks like ovarian hyperstimulation, paving the way for tailored interventions that could enhance fertility preservation and overall patient care.
https://www.nature.com/articles/s41591-025-03984-1
Finally, on the policy front, the Trump administration advanced initiatives to expand access to infertility treatments, including efforts to reduce costs for fertility medications and encourage employer-based coverage options. Notably, there was increased attention to Restorative Reproductive Medicine (RRM), an approach supported by conservative and Catholic-aligned groups that emphasizes identifying and treating root causes of infertility through holistic methods, lifestyle interventions, and natural fertility restoration—rather than relying primarily on artificial techniques like IVF. This alignment with promoting natural conception resonates deeply with emerging paradigms that prioritize underlying health restoration, offering a complementary pathway for patients seeking ethical and root-cause-focused solutions.
https://www.wsj.com/opinion/want-babies-treat-infertilitys-root-causes-ivf-rrm-boom-trump-fe49bb4b
These developments underscore a dynamic year of innovation and policy evolution in reproductive health, bringing hope to countless individuals and families.
Looking Ahead to 2026 with Renewed Commitment
As we step into 2026, infuse Life Science, Inc. is poised for an exciting phase of growth and impact. We are committed to transitioning our innovative medical device into full-scale production development, ensuring they reach patients and healthcare providers worldwide.
To uphold the standards of quality and safety, we will advance our development processes in compliance with QMSR (Quality Management System Regulation) and QMS (Quality Management System) requirements, with the goal of achieving ISO 13485 certification—a milestone that will strengthen our global credibility and facilitate broader market access.
Furthermore, we aim to deepen collaborations with key opinion leaders across reproductive and cardiovascular medicine on an international scale, fostering knowledge exchange and innovation. Building on this momentum, we will further expand and strengthen our consulting business, delivering even greater value to our clients through cutting-edge expertise and successful project outcomes.
With gratitude for your continued partnership and inspired by the remarkable advancements in reproductive medicine throughout 2025, we look forward to a year of meaningful progress, ethical innovation, and lasting contributions to patient care.
Wishing you a prosperous and joyful 2026.
Dai Saito, CEO.
infuse Life Science, Inc.
謹賀新年 2026
新年を迎え、インフューズライフサイエンス株式会社一同より、ステークホルダーの皆様、パートナーの皆様、クライアントの皆様、そしてこれまで支えてくださった全ての支援者の皆様に、心より新年のご挨拶と感謝を申し上げます。2025年も変わらぬご信頼とご協力をいただき、誠にありがとうございました。
昨年は、当社の中核事業において目覚ましい進展と成果を上げた一年となりました。不妊の早期発見を目指す革新的な診断機器については、試作開発フェーズを無事完了し、近日中にプレスリリースにてその成果を発表できる運びとなりました。また、治療用医療機器の開発も順調に進んでおり、1月には主要な成果を助成金事業ご採択元へご報告できる見込みです。
コンサルティング事業におきましても、皆様のご支援のおかげで大きな成長を遂げました。現在、4社のクライアントに対し6つのプロジェクトを推進しており、それぞれにおいて顕著な成果を上げております。
循環器領域では、初めてGREATシンポジウムに賛助参加するというマイルストーンを達成し、欧米の救急・集中治療分野のキーオピニオンリーダー(KOL)への認知が大幅に向上しました。
2025年生殖医療分野のホットトピック
2025年の生殖医療分野は、不妊に悩む患者さんの診断・治療・予後を大きく変革する可能性を秘めた、数々の画期的な進展が見られました。
最も注目すべき進歩の一つは、AIによる胚選択システムの進化です。数百万枚のタイムラプス胚画像で学習された基礎モデル「FEMI」は、従来の手動グレーディングや従来のAIモデルを上回る性能を示し、染色体正常性(euploidy)、胚盤胞の質、その他の重要指標を高精度に予測しました。この非侵襲的な技術は、着床可能性の評価精度を飛躍的に向上させ、着床率の改善、生児獲得率の上昇、流産リスクの低減を実現する可能性があり、IVFサイクルにおける胚選択のより標準化された効率的な手法を提供します。
もう一つの大きなブレークスルーは、多嚢胞性卵巣症候群(PCOS)に関する理解の深化です。不妊の主要原因であるPCOSについて、大規模国際コホートにおける臨床変数のデータ駆動型クラスタリングにより、4つの明確なサブタイプ(高アンドロゲン型、肥満関連型、高SHBG型、高LH/AMH型)が同定されました。これらのサブタイプは、代謝リスク、生殖予後、合併症において異なり、より個別化された診断・治療戦略を可能にします。例えば、あるサブタイプでは生児獲得率が高く、別のサブタイプでは卵巣過剰刺激症候群のリスクが高いことが示され、個別化された介入により妊孕性温存と患者ケア全体の向上が期待されます。
最後に、政策面では、トランプ政権が不妊治療へのアクセス拡大に向けた取り組みを推進し、治療薬のコスト低減や企業による保険適用促進を図りました。特に注目されたのは、Restorative Reproductive Medicine(RRM:修復生殖医療)への関心の高まりです。保守派やカトリック系団体が支持するこのアプローチは、ホリスティックな手法、生活習慣介入、自然な妊孕性回復を通じて不妊の根本原因を特定・治療することに重点を置き、IVFなどの人工的な手法に過度に依存しないことを特徴とします。自然妊娠を促進するこの方向性は、根本的な健康回復を優先する新たなパラダイムと深く共鳴し、倫理的かつ根因治療を求める患者さんに補完的な選択肢を提供します。
これらの進展は、生殖医療分野におけるイノベーションと政策のダイナミックな進化を示すものであり、多くの個人と家族に希望をもたらしました。
2026年への抱負
2026年を迎え、インフューズライフサイエンス株式会社はさらなる成長とインパクトのステージに立っています。診断用医療機器を量産開発フェーズへと移行させることに全力で取り組んでまいります。これにより、世界中の患者さんや医療従事者の皆様のお手元に製品をお届けできるようにいたします。
品質と安全性の水準を維持するため、QMSR(Quality Management System Regulation)およびQMS(Quality Management System)省令に準拠した開発プロセスを推進し、ISO 13485認証の取得を目指します。このマイルストーンは、当社のグローバルな信頼性を強化し、より広い市場アクセスを実現するものです。
さらに、生殖医療および循環器医療の国際的なキーオピニオンリーダーとの連携を深め、知識交換とイノベーションを促進します。この勢いを活かし、コンサルティング事業もさらに拡大・強化し、最先端の専門性とプロジェクト成功により、クライアントの皆様にさらなる価値を提供してまいります。
2025年の生殖医療の目覚ましい進展に触発され、皆様とのパートナーシップに感謝しつつ、2026年は意義深い進歩、倫理的なイノベーション、そして患者ケアへの持続的な貢献を実現する一年となることを楽しみにしております。
皆様のご多幸とご発展を心よりお祈り申し上げます。
齋藤大介, 代表取締役CEO
インフューズライフサイエンス株式会社
infuse Life Science, Inc. today announced that it has successfully declared the “SECURITY ACTION” One Star level, an information security self-declaration program promoted by the Information-technology Promotion Agency (IPA) of Japan.
This declaration signifies iLS's commitment to implementing the fundamental "Information Security 5 To-dos" (情報セキュリティ5か条) recommended by the IPA. By making this public commitment, we aim to ensure the safe and reliable handling of information for our customers and business partners.
Time truly flies! We are thrilled to announce the celebration of infuse Life Science's 1st anniversary on October 1st. We are steadily approaching the realization of the vision we promised that day: to use the power of life science to solve Japan and the world's demographic challenges, particularly the declining birthrate.
This report details our progress over the nine months leading up to this milestone.
1. Research & Development in Ob-Gyn
The planned functional prototype has been successfully completed and is now in excellent operational condition. We are developing a specialized, multi-product lineup focused on the vital yet often underfunded field of Ob-Gyn—a market deemed small but highly significant, often overlooked by larger corporations.
This prototype represents a crucial step in our strategy to expand our offerings across the entire clinical pathway: observation, examination, diagnosis, and ultimately, treatment.
Based on this successful result, we have initiated collaboration discussions with several key investors and medical device manufacturers, receiving overwhelmingly positive feedback and strong indications of interest from nearly all parties. The next phase involves transitioning to final product development, operating under a robust QSR/QMS/ISO13485 level.
2. Consulting Business
As detailed on the Services page of our website, our business model education and consulting services continue to be highly regarded. We encourage you to read the testimonials and contact us today. We promise to provide proposals—in terms of content and pricing—that are precisely aligned with your company's specific needs and demands.
3. Staff Recruitment
As we enter this critical new phase of growth and development, we are actively seeking passionate staff members. Join us in the fight against global and domestic demographic challenges, leveraging cutting-edge medical devices to address the declining birthrate. We need your skills and dedication to stand with us.
While we enjoy the arrival of autumn, we are also mindful of the intense spread of infectious diseases. We wish everyone good health and continued well-being.
The devices are not approved as medical device. These are aiming at introducing our business to the investors, but not intend to show to the medical professionals or patients.